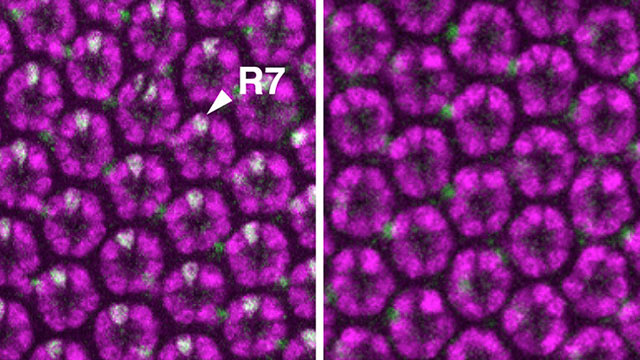
The optical units in a fruit fly’s eye make a mosaic pattern. A common gene mutation damages R7 cells, which allow fruit flies to see ultraviolet light.
Just by slowing their metabolism, mutant fruit flies can go from zero to hero.
In a new Northwestern University study, researchers slowed mutant fruit flies’ metabolic rates by 50%, and the expected detrimental effects of many mutations never manifested. After experimentally testing fruit flies’ many different genetic mutations, the researchers found the same result each time.

Richard Carthew
“When the flies developed at a normal rate, developmental problems occurred,” said Northwestern’s Richard Carthew, who led the experimental research. “When we slowed the rate, developmental problems disappeared. They develop slower and grow slower, but, otherwise, they are normal animals.”
“This upends the paradigm of everything we know about development,” added Northwestern’s Luís Amaral, who led the computational research. “We have always thought that if you ‘break’ some genes, there will be serious developmental consequences. It turns out that’s not true for some genes — as long as you also slow the metabolism of the growing organism.”
The research could explain a number of factors, such as why factory-farmed chickens that are bred for hyper growth have more developmental problems or why caloric restriction is linked to longevity.
The study published today (July 25) in the journal Cell. Carthew is a professor of molecular biosciences in Northwestern’s Weinberg College of Arts and Sciences and professor of biochemistry and molecular genetics in the Feinberg School of Medicine. Amaral is the Erastus O. Haven Professor of Chemical and Biological Engineering in Northwestern’s McCormick School of Engineering.

Luis Amaral
In perhaps the study’s most striking discovery, researchers found that fruit flies — with slowed metabolic rates — could live without any microRNAs, which was previously thought impossible. Found in all plant and animal species, microRNAs play a key role in regulating gene expression. To put it simply: microRNAs control development, physiology and behavior.
“We know from 20 years of research, microRNAs are essential for life. If you didn’t have any microRNAs, you would be dead. Simple as that,” Carthew said. “In our study, we slowed down the metabolism of fruit flies that were not making any microRNAs whatsoever. They survived, they grew and they became normal adults.
“Our result concludes that this entire family of gene regulators is not essential,” he added. “All you have to do is slow metabolism by roughly 50%.”
Nobel laureate Thomas Hunt Morgan first noted the connection between diet and genetic mutations in 1915. When he raised mutant fruit flies on limited amounts of poor food, Morgan noticed that some mutations were never expressed.
“He thought it was interesting, but he had no explanation,” Carthew said.
Carthew and Amaral now believe the answer is feedback control. Common in biology, engineering, economics and many other fields, feedback control enables complex systems to adjust performance in order to meet a desired response. After completing hundreds of experiments across several years, the Northwestern duo believes that a slower metabolism gives the animals’ systems time to correct errors.
This upends the paradigm of everything we know about development.
“When you look at all the different proteins and genes that interact within a cell, you can get overwhelmed by all the components and the interactions among them,” Amaral said. “If you are growing fast and something goes wrong, it can be catastrophic. You need these complex networks because they increase redundancy to prevent catastrophe.
“But if you are growing slowly, you might not need such a complex system,” he said. “You have more time to adjust to mistakes and react to changes.”
In other words, if you give the system more time, it will eventually get to where it needs to be. Carthew, who is also a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, said this finding could eventually be applied to cancer.
“Tumors are extremely metabolically active,” he said. “Tumors soak up an enormous amount of energy, which is why cancer patients are often exhausted. We could potentially think about ways to target the metabolism of cancer cells. Maybe by slowing their metabolic rate, we could stop the oncogenic mutations in tumor cells from expressing their cancer phenotype.”
The study, “Repressive gene regulation synchronizes development with cellular metabolism,” was supported by the Chicago Biomedical Consortium, the Malkin Foundation, the Rappaport Foundation, the National Science Foundation (award number 1764421), the Simons Foundation (award number 597491) and the National Institutes of Health (award numbers T32 GM008061, T32 CA080621, P50 GM81891 and R35 GM118144). Northwestern Ph.D. students Justin J. Cassidy and Sebastian M. Bernasek are the paper’s co-first authors.
This post was originally published on July 25th, 2019.
